Excipient MultiFunctional A
1 of 4 products in this brandExcipient MultiFunctional A is a versatile hypromellose, compliant with USP, JP, EP. The low viscosity grades are especially suitable for the design of film-coated tablet formulations. Excipient MultiFunctional A is effective as a binder, since it does not interact with drugs. It has superior stability, a non-ionic character and is widely used as a binder for granulation. Due to its plant derived nature and excellent moisture stability, Excipient MultiFunctional A replaces gelatin in hard capsule manufacturing.
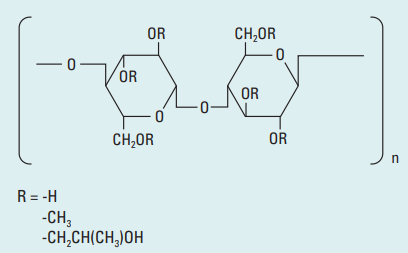
Chemical Name: Hydroxypropyl Methylcellulose
Functions: Binder, Controlled Release Agent, Sustained Release Agent, Thickener, Viscosity Modifier
Dosage Form: Suspension, Tablets
Certifications & Compliance: European Pharmacopeia (Ph. Eur.), GMP, Japan Pharmacopeia (JP), United States Pharmacopeia (USP)
Enhanced TDS
Identification
- Chemical Name
Applications
- Dosage Form
- Manufacturing Technology
Applications & Uses
- Product Applications
-
- Suitable for thickening
- Suitable for suspending
- Liquid and semi-solid
- Suitable for sustained release
Features & Benefits
- HPMC Key Quality Attributes in Matrix Tablets
-
- Decrease in initial erosion.
- Slower drug release.
- No effect on tablet hardness.
Functions
- Pharma & Nutraceuticals Functions
- Function
-
Controlled Release Agent, Viscosity Modifier, Sustained Release Agent, Thickener, Binder
Identification & Functionality
- Structure
-
Packaging & Availability
- Packaging Type
- Packaging Information
-
- Fiber drum (40-50 kg) with polyethylene single bag inside of 450 mm x h 740 mm (110 liters).
- Big bag of 1060 x w 710 x h 1250 mm (up to 1040 liters).
Properties
- Physical Form
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- Grade
- Regulatory Information
-
- EU-Regulations
- No REACH Annex XVII restrictions. This product is not on the REACH Candidate List
- This product is not on the REACH Annex XIV List
- This product Safety Data Sheet according to Regulation (EC) No. 1907/2006 (REACH) with its amendment Regulation (EU) 2020/878 SDS No: 10896-0310 16/03/2021 (Revision date) GB - en 8/9
- This product is not subject to Regulation (EU) No 649/2012 of the European Parliament and of the Council of 4 july 2012 concerning the export and import of hazardous chemicals. This product is not subject to Regulation (EU) No 2019/1021 of the European Parliament and of the Council of 20 June 2019 on persistent organic pollutants
- Contains no substance subject to Regulation (EU) 2019/1148 of the European Parliament and of the Council of 20 June 2019 on the marketing and use of explosives precursors.
- National regulations
- Listed on the United States TSCA (Toxic Substances Control Act) inventory
- Listed on the Canadian DSL (Domestic Substances List)
- Listed introduction on Australian Industrial Chemicals Introduction Scheme (AICIS Inventory)
- Listed on the EEC inventory EINECS (European Inventory of Existing Commercial Chemical Substances)- Directive 79/831/EEC, sixth Amendment of Directive 67/548/EEC (dangerous substances)
- Listed on the Japanese ENCS (Existing & New Chemical Substances) inventory Listed on PICCS (Philippines Inventory of Chemicals and Chemical Substances)
- Listed on NZIoC (New Zealand Inventory of Chemicals)
- Listed on the TCSI (Taiwan Chemical Substance Inventory) Listed on IECSC (Inventory of Existing Chemical Substances Produced or Imported in China) Listed on KECL/KECI (Korean Existing Chemicals Inventory)
Storage & Handling
- Storage Information
-
- Keep dry. Store away from excess heat and sunlight.
- Store preferentially in original packaging.
